ABSTRACT
Bariatric surgery can result in substantial weight loss and significant metabolic improvements. Therefore, postoperatively, clinicians should be prepared to taper treatments for weight-related chronic metabolic diseases. For patients with type 2 diabetes, early and dramatic improvements in glucose homeostasis require anticipatory management. This includes insulin dose reductions, discontinuation of certain oral agents, and close monitoring. Antihypertensive medications should be adjusted to avoid hypotension. Even after postoperative improvements in dyslipidemia, some patients will continue to meet criteria for statin therapy. While many obesity-related diseases will improve, clinicians should also be prepared to manage postoperative medical and nutritional complications. Micronutrient deficiencies are common, and professional guidelines provide recommendations for preoperative screening, universal postoperative supplementation, micronutrient monitoring, and repletion strategies. Changes in gastrointestinal physiology may result in dumping syndrome, and patients may report early gastrointestinal and vasomotor symptoms after eating. In contrast, post-bariatric hypoglycemia is a rare complication of malabsorptive procedures, resulting in insulin-mediated hypoglycemia after carbohydrate-containing meals. Rapid weight loss may increase the risk of cholelithiasis, which can be mitigated by ursodiol. After malabsorptive procedures, enteric hyperoxaluria and other factors may result in nephrolithiasis, which can be addressed with hydration, dietary interventions, and calcium. All bariatric operations induce a high bone turnover state, with declining bone mineral density (BMD) and increased fracture risk. Appropriate strategies include adequate calcium and vitamin D supplementation and targeted BMD screening. Long-term strategies to prevent weight regain include adherence to comprehensive lifestyle interventions and identification and avoidance of medications that promote weight gain and may include weight management medications. In summary, given dramatic physiologic changes with bariatric surgery, clinicians should be prepared to taper treatments for chronic metabolic diseases, to manage postoperative medical and nutritional complications, and to identify and manage risk for weight regain. For complete coverage of all related areas of Endocrinology, please visit our on-line FREE web-text, WWW.ENDOTEXT.ORG.
POSTOPERATIVE APPROACH TO CHRONIC METABOLIC CONDITIONS
In the perioperative and early postoperative periods (usually the first 30 to 90 days after surgery), a patient’s surgeon will monitor closely for surgical complications such as anastomotic leak, deep vein thrombosis, and infection. An experienced dietitian generally assists with meal initiation and progression. Later, regular follow-up with the surgeon—including, eventually, annual follow-up for life—is essential for assessing weight loss success and reinforcing necessary lifestyle modifications. The primary care provider or endocrinologist assumes responsibility for the early and later postoperative management of chronic medical conditions, including diabetes, hypertension, and dyslipidemia. This section summarizes the effects of bariatric surgery on those conditions and the recommended approach to management.
Postoperative Diabetes Management
Bariatric surgery dramatically improves glucose homeostasis and type 2 diabetes (T2D). After RYGB in particular, these improvements are both weight loss-dependent and weight loss-independent, with weight loss-independent effects likely mediated by alterations in gut hormones, gastrointestinal tract nutrient sensing, bile acid metabolism, and the gut microbiome (6,7). Due to these complex factors and the effects of postoperative calorie restriction, improvement in glucose homeostasis is evident within days to weeks following RYGB. (8,9). In an early systematic review and meta-analysis, diabetes remission was observed in 99% of those with T2D who underwent BPD/DS, 84% of those who underwent RYGB, and 48% of those who underwent AGB (1). Of participants in the Longitudinal Assessment of Bariatric Surgery-2 (LABS-2) study with T2D, 59% of RYGB participants and 25% of AGB participants were in diabetes remission 7 years after surgery (10). Even after controlling for differences in the amount of weight lost, the diabetes remission rate after RYGB was almost double that after AGB (11). SG is positioned between RYGB and AGB in T2D effectiveness (12-14).
The hospitalist, endocrinologist, or primary care provider caring for a bariatric surgery patient with T2D must anticipate a quick and potentially dramatic improvement in glycemic status (15,16). Typically, oral insulin secretagogues (sulfonylureas and meglitinides) are discontinued at the time of surgery to decrease hypoglycemia risk. Insulin doses should be reduced in the hospital and upon discharge home, with strict instructions provided to the patient for self-monitoring blood glucose levels and adjustments of insulin doses to avoid hypoglycemia. Metformin is often continued postoperatively, with appropriate caution exercised in patients with reduced kidney function, until blood glucose levels and hemoglobin A1c in the subsequent months suggest that it can be discontinued. While incretin-based therapies (GLP-1 receptor agonists and DPP-4 inhibitors) can generally be continued safely (after temporarily holding GLP-1 receptor agonists for the procedure itself per hospital protocol), they are often discontinued postoperatively because of the apparent effects of bariatric surgery on incretin physiology. SGLT2 inhibitors are likely not needed for glycemic control postoperatively but may be continued in some individuals with other (e.g., heart failure or chronic kidney disease) indications. Thiazolidinediones could also be theoretically continued but are often discontinued in part due to expected postoperative changes in insulin sensitivity and volume status. Alpha-glucosidase inhibitors should be discontinued due to their gastrointestinal effects.
Close glucose monitoring is critical regardless of the initial postoperative T2D medication regimen. For patients using insulin or an insulin secretagogue, this must include patient self-monitoring of blood glucose levels with a clear plan for adjustments. For others, self-monitoring may be reassuring and should be individualized. Hemoglobin A1c monitoring should be routinely continued long-term (years). While glucose control improves to the point of complete remission in most patients in the year after bariatric surgery (70% or more (10) depending on the procedure), certain patients are at higher risk for not achieving remission or for having diabetes recur over time, including older patients, those with a longer-duration of diabetes, and those who were using insulin or required more than one non-insulin medication (11,17). Such patients are characterized by a greater impairment in insulin secretory capacity. Long-term data elucidate the proportions of T2D patients who achieve and maintain complete remission: In a cohort of RYGB patients, of those with T2D preoperatively, 75% had remitted 2 years postoperatively, 62% at 6 years, and 51% at 12 years (17). In the LABS-2 study, 7 years after surgery, 60% of RYGB participants and 20% of AGB participants were in diabetes remission (10).
In patients not reaching glycemic targets or experiencing relapses, diabetes therapies can be resumed or added. A reasonable approach is first to add/restart metformin, then, if needed, add one or more other weight-neutral or weight loss-promoting agents such as a GLP-1 receptor agonist, a DPP-4 inhibitor, or an SGLT2 inhibitor.
Post Opérative Hypertension Management
Reductions in systolic and diastolic blood pressure have been demonstrated at just one week after RYGB. (18), suggesting weight loss-dependent and weight loss-independent mechanisms (19). An early systematic review and meta-analysis of bariatric surgery outcomes demonstrated that, of patients with a preoperative diagnosis of hypertension, hypertension resolved completely after surgery in 62% and resolved or improved in 79% (1). Frank remission was observed in 83% of those who underwent BPD/DS, 68% of those who underwent RYGB, and 43% of those who underwent AGB. Subsequent studies have yielded less impressive but still very favorable results (19,20). For example, of participants in the LABS-2 study with hypertension, 38% of RYGB participants and 17% of AGB participants had complete remission of hypertension 3 years after surgery (21), and 33% of RYGB participants and 17% of AGB participants had complete remission after 7 years (10). SG also has a substantial effect on hypertension, with resolution or improvement in the majority of cases (22), although the odds of resolution of hypertension appear greater after RYGB than SG (23).
Because the effect of bariatric surgery on blood pressure is thought to be variable and potentially less durable than on glucose metabolism, the Clinical Practice Guidelines of the American Association of Clinical Endocrinologists (AACE), The Obesity Society (TOS), and American Society for Metabolic and Bariatric Surgery (ASMBS) recommend against the preemptive discontinuation of antihypertensive medications (24). Instead, providers should pay close attention to blood pressure postoperatively in the hospital and at every postoperative clinic visit and adjust medications when indicated (15).
Postoperative Dyslipidemia Management
Bariatric surgery may improve dyslipidemia by altering diet, various endocrine and inflammatory factors, bile acid metabolism, and potentially even the intestinal microbiome (25). An early systematic review and meta-analysis of bariatric surgery outcomes demonstrated that among patients undergoing AGB, RYGB, gastroplasty, or BPD/DS, hyperlipidemia improved in 79%, hypercholesterolemia improved in 71%, and hypertriglyceridemia improved in 82% (1). Of participants in the Longitudinal Assessment of Bariatric Surgery-2 (LABS-2) study, 62% of RYGB participants and 27% of AGB participants had remission of dyslipidemia 3 years after surgery (21), and percentages were generally similar 7 years after surgery (10). Regarding the SG, a systematic review confirmed its effectiveness for the treatment of dyslipidemia (26). In STAMPEDE, a randomized controlled trial (RCT) of RYGB, SG, or intensive medical therapy alone among overweight and obese patients with T2D, both RYGB and SG increased HDL and decreased TG levels compared to placebo (13). Change in LDL level was not different between groups, although the number of medications needed to treat hyperlipidemia was lower in the surgical groups than in the medical therapy group.
Unlike insulin and anti-hypertensive medications, which must be decreased or discontinued when no longer needed to mitigate the acute dangers of overtreatment, lipid-lowering medications may be continued during the metabolically dynamic early postoperative period. Moreover, even after postoperative improvement in dyslipidemia, many bariatric surgery patients will continue to meet criteria for statin use based on the current American College of Cardiology/American Heart Association guideline (27) and US Preventive Services Task Force recommendations (28), especially those at very high risk for cardiovascular events, including secondary prevention. With this in mind, for many patients, endocrinologists and primary care providers should be cautious about creating expectations that statin therapy will be discontinued postoperatively. Instead, a patient’s cardiovascular risk should be periodically evaluated and the potential role of statins discussed in an individualized manner.
Medication Adjustments
Nonsteroidal anti-inflammatory drugs (NSAIDs) should be avoided after bariatric surgery because of the risk of gastric and marginal ulcer development (29). In many bariatric centers, proton pump inhibitor therapy is prescribed postoperatively, as evidence from cohort studies suggests that it may decrease ulcer risk (30), and in light of the potential for incident or worsening gastroesophageal reflux disease (GERD) after SG (31). Endocrinologists and primary care providers should be prepared to make adjustments to the dose of any medication that is dosed based on weight (e.g., levothyroxine), and to consider potential effects of malabsorption on a patient’s usual oral medications.
PREVENTION AND TREATMENT OF POSTOPERATIVE MEDICAL AND NUTRITIONAL COMPLICATIONS
While a patient’s surgeon monitors closely for postoperative surgical complications, the primary care provider or endocrinologist often identifies and manages postoperative medical and nutritional complications. This section reviews these potential complications (Table 1), with attention to pathophysiology, screening, and therapeutic approach.
Micronutrient Deficiencies
Given the dietary changes, rerouting of nutrient flow, and gut anatomy/physiology alterations that occur after bariatric surgery, patients who undergo these procedures are at risk for micronutrient deficiencies. Some of these deficiencies can result in severe consequences, such as neuropathy, heart failure, and encephalopathy. Therefore, it is essential that patients comprehend the importance of compliance and the need for lifelong supplementation. Patients who have malabsorptive procedures, such as RYGB or BPD/DS, are at highest risk for micronutrient deficiencies, but even with restrictive procedures, decreased oral intake and poor tolerance to certain food groups may also increase the risk for micronutrient deficiencies. Therefore, preoperative nutritional evaluation and postoperative monitoring and supplementation are necessary.
Tables 2-5 represent recommendations that have been adapted and modified from the American Society for Metabolic and Bariatric Surgery (ASMBS) Integrated Health Nutrition Guidelines (32) and the Clinical Practice Guidelines from the combined American Association of Clinical Endocrinologists (AACE), The Obesity Society (TOS), and ASMBS (24). These recommendations for adults reflect general guidelines, and patients with specific diseases may require further evaluation and closer monitoring. For example, nutritional anemias resulting from malabsorptive bariatric surgical procedures in the setting of appropriate iron repletion might also involve other micronutrient deficiencies in vitamin B12, folate, protein, copper, selenium, and zinc, and these should be evaluated.
Preoperative micronutrient screening recommendations are listed in Table 2. Ideally, preexisting micronutrient deficiencies would be corrected prior to surgery in order to avoid clinically symptomatic or severe disease. Suboptimal levels of 25-hydroxyvitamin D are particularly common and may require supplementation prior to surgery (33).
Table 2.
Preoperative Micronutrient Screening Recommendations
| Micronutrient | Surgical population | Screening laboratory test (optional tests) |
|---|---|---|
| Vitamin B12 (cobalamin) | All | Vitamin B12 (optional: MMA) |
| Folate (folic acid) |
All | Folate (optional: RBC folate, homocysteine, MMA) |
| Iron | All | Iron, TIBC, ferritin |
| Vitamin D | All | 25-hydroxyvitamin D |
| Calcium | All | Calcium (optional: intact PTH, 24-hour urinary calcium) |
| Thiamine | All | Thiamine |
| Vitamin A | RYGB, BPD/DS | Vitamin A |
| Vitamin D | RYGB, BPD/DS | Vitamin E |
| Zinc | RYGB, BPD/DS | Zinc |
| Copper | RYGB, BPD/DS | Copper and ceruloplasmin |
-
Table modified from the ASMBS Integrated Health Nutritional Guidelines (32).
Universal postoperative supplementation (Table 3) is an important component of postoperative care. For example, vitamin B12 deficiency is common after RYGB without adequate supplementation, and oral doses of 350 mcg/day have been shown to maintain normal plasma B12 levels. Other suggested micronutrient doses are either based on expert opinion or are similar to the recommended dietary allowance (RDA). These universally supplemented micronutrients are usually obtained from a bariatric, high-potency multivitamin.
Table 3.
Recommended Postoperative Supplementation of Vitamins and Minerals
| Micronutrient | Supplementation | |
|---|---|---|
| Usually within a multivitamin with minerals product | ||
| Thiamine | At least 12 mg/day, preferably 50-100 mg/day | |
| Vitamin B12 (cobalamin) | Oral or sublingual: 350-500 mcg/day Intramuscular: 1000 mcg/month Titrate to normal levels |
|
| Folate (folic acid) | 400-800 mcg/day Women of childbearing age: 800-1000 mcg/day |
|
| Iron | 18 mg/day elemental iron RYGB, SG, BPD/DS or menstruating women: 45-60 mg/day Take separately from calcium supplements |
|
| Vitamin D | D3 3000 IU/day Titrate to 25-hydroxyvitamin D level at least 30 ng/mL |
|
| Vitamin A | AGB: vitamin A 5000 IU/day RYGB or SG: vitamin A 5,000-10,000 IU/day BPD/DS: vitamin A 10,000 IU/day |
|
| Vitamin E | 15 mg/day | |
| Vitamin K | AGB, SG or RYGB: 90-120 mcg/day BPD/DS: 300 mcg/day |
|
| Zinc | SG or AGB: 8-11 mg/day RYGB: 8-22 mg/day BPD/DS: 16-22 mg/day |
|
| Copper | SG or AGB: 1 mg/day RYGB or BPD/DS: 2 mg/day |
|
| As separate supplementation | ||
| Calcium | AGB, SG, RYGB: calcium 1200-1500 mg/day (diet + supplements) BPD/DS: calcium 1800-2400 mg/day (diet + supplements) (as calcium citrate, in divided doses) |
|
-
Table modified from the ASMBS Integrated Health Nutritional Guidelines (32).
Most micronutrients are provided in multivitamins, and chewable multivitamins are recommended postoperatively. Bariatric surgery-specific, high-potency multivitamins are available and often contain the recommended doses of micronutrients in one tablet daily. Alternatively, multivitamins for the general population can be used, provided that attention is paid to the product’s micronutrient contents. If using general (not bariatric, high potency) multivitamins, the ASMBS recommends one general multivitamin tablet daily for patients who have had AGB, or 2 general multivitamin tablets daily for those undergoing SG, RYGB or BPD/DS.
Multivitamins do not contain the recommended doses of calcium, because calcium can impede the absorption of other micronutrients. Therefore, separate calcium supplementation is usually required. Calcium citrate is the preferred form of supplemental calcium, as it is better absorbed than calcium carbonate in the state of impaired gastric acid production. A patient’s dietary calcium intake should be considered when determining the dose of a calcium supplement, as the recommended intakes are generally total daily intakes (diet plus supplements). Iron absorption may be enhanced by co-administration of vitamin C (500-1000 mg) to create an acidic environment or when taken with meat. If inadequate absorption or intolerance occurs, parenteral iron replacement may be necessary.
A suggested schedule for postoperative biochemical monitoring is listed in Table 4. Patients who develop micronutrient deficiencies may need more frequent monitoring.
Table 4.
Schedule for Postoperative Micronutrient Monitoring
| 6 months | 12 months | 18 months | 24 months | Annually | |
|---|---|---|---|---|---|
| Vitamin B12 | X | X | X | X | X |
| Folate | X | X | X | X | X |
| Iron, ferritin | X | X | X | X | X |
| 25-hydroxyvitamin D | X | X | X | X | X |
| Calcium | X | X | X | X | X |
| Intact PTH | X | X | X | X | X |
| 24-hour urinary calcium | X | X | X | X | |
| Vitamin A | X | X | X | ||
| Thiamine, copper, zinc, selenium | (optional, or if specific findings or concomitant deficiencies) | ||||
Examinations should be performed after RYGB or BPD/DS. All of these could be suggested for patients submitted to restrictive surgery where frank deficiencies are less common.
Some surgeons perform additional early biochemical evaluation 3 months postoperatively, and the AACE/TOS/ASMBS Clinical Practice Guidelines suggest evaluation earlier than 6 months for some micronutrients (24).
Oral repletion is often sufficient for correcting micronutrient deficiencies, although parenteral therapy may be required in severe disease. After a repletion course, biochemical testing should be performed, and a maintenance dose should be established. Micronutrient deficiencies may co-exist; for example, malabsorptive procedures may result in deficiencies of the fat-soluble vitamins A, E and K.
Table 5.
Repletion Recommendations for Individuals Diagnosed with Micronutrient Deficiencies
| Micronutrient | Repletion recommendation |
|---|---|
| Thiamine | Oral: 100 mg 2-3 times daily IM: 250 mg daily for 3-5 days or 100-250 mg monthly IV: 200 mg 2-3 times daily to 500 mg 1-2 times daily for 3-5 days, followed by 250 mg/day for 3-5 days Severe disease: administer thiamine prior to dextrose-containing solutions |
| Vitamin B12 (cobalamin) | Oral: 1000 mcg/day |
| Folate (folic acid) | 1000 mcg/day orally once, then resume daily maintenance dose |
| Iron | 150-200 mg elemental iron/day, up to 300 mg 2-3 times daily Calcium may impair iron absorption Consider co-administration of vitamin C to enhance absorption Consider IV iron infusions for severe/refractory iron deficiency |
| Vitamin D | D3 6000 IU/day or D2 50,000 IU 1-3 times per week, or more if needed to achieve and maintain 25-hydroxyvitamin D >30 ng/mL |
| Calcium | Increase dose and titrate to normalize PTH ± 24-hr urinary calcium level* |
| Vitamin A | 10,000-25,000 IU/day orally until clinical improvement (1-2 weeks) With corneal changes: 50,000-100,000 IU IM x 3 days, then 50,000 IU/day IM for 2 weeks |
| Vitamin E | Optimal therapeutic dose not clearly defined, consider 100-400 IU/day |
| Vitamin K | Acute malabsorption: 10 mg parentally Chronic malabsorption: 1-2 mg/day orally or 1-2 mg/week parentally |
| Zinc | There is insufficient evidence to make a dose-related recommendation |
| Copper | Mild-moderate deficiency: oral copper gluconate or sulfate 3-8 mg/day Severe deficiency: 2-4 mg/day of intravenous copper x 6 days |
-
Table modified from the ASMBS Integrated Health Nutritional Guidelines (32).
-
IM, intramuscular; IV, intravenous.
- †
-
Recommendation from the AACE/TOS/ASMBS Clinical Practice Guidelines (24).
- *
-
In chronic kidney disease, PTH goal should be appropriate for renal function (34,35).
Dumping Syndrome and Post-Bariatric Hypoglycemia
Early and late dumping syndromes are a result of altered gastrointestinal anatomy and hormone secretion after bariatric surgery. The two syndromes have distinct symptomatology and pathophysiology though there is considerable overlap in dietary triggers and treatment approaches. Late dumping syndrome is hallmarked by hypoglycemia and will henceforth be referred to as post-bariatric hypoglycemia (PBH).
Early Dumping Syndrome
Early dumping syndrome (DS) typically occurs within 1 hour of eating and is characterized by both gastrointestinal (nausea, abdominal fullness, diarrhea) and vasomotor symptoms (fainting, sleepiness, weakness, diaphoresis, palpitations, and desire to lie down) (36). Dumping syndrome symptoms can appear as early as 6 weeks after surgery and has been reported to affect up to 20% according to large survey studies and up to 40% in smaller prospective studies of individuals who have undergone both restrictive and malabsorptive procedures (37-40). The pathophysiology of DS is not completely understood but is thought to be due to both a rapid delivery of nutrients to the small intestine causing an osmotic shift of intravascular fluid to the intestinal lumen as well as an increased release of gastrointestinal hormones that disrupt motility and hemodynamic status (41-43). The rate of DS may be more common in those who underwent cholecystectomy suggesting a contribution of altered bile acid release (44). There is debate in the literature on whether DS is an adaptive consequence of bariatric surgery that helps restrict food intake and aids weight loss versus an adverse consequence that reduces quality of life and does not contribute to weight loss (37,45,46).
The diagnosis of DS should be made after the exclusion of more serious entities such as intestinal fistulas, adhesions, ischemia, herniation, obstipation, and gallstone disease which may have shared clinical features (42). There are validated questionnaires as well as provocation tests that have been used to confirm DS in research settings. Oral glucose challenge with an increase in heart rate and hematocrit (indicating hemoconcentration) is one such approach (36,47,48).
The first line treatment for DS is to modify the diet to avoid foods that worsen symptoms (oftentimes calorie-dense foods with high fat/refined sugar content and low in fiber), eating small volume meals, not eating and drinking at the same time, eating slowly, chewing well, and avoiding alcohol. Indeed, patients often implement these changes on their own and, over time, symptom severity improves or resolves in many (if not most) patients. In addition, lying down for 30 minutes after eating to slow gastric emptying and mitigate symptoms of hypovolemia may be helpful if symptoms occur (49). There are several small interventional studies and case reports that support the use of dietary supplements (e.g., pectin, guar gum) that increase food viscosity and reduced symptoms of DS, however low palatability and potential choking hazard and bowel obstruction are downsides to their use (42). Somatostatin analogs have also been tested in small studies, although this class of drugs are expensive, involve subcutaneous or intramuscular injections, and have gastrointestinal side effects (42). Enteral tube feedings or bariatric surgery reversal have been reported to improve symptoms when all else fails (42).
Post Bariatric Hypoglycemia
Post-bariatric hypoglycemia (PBH) is a rare complication of bariatric surgery that occurs several months to years after procedures that rapidly pass nutrients through the stomach (or stomach remnant) directly to the small intestine and has not been reported with restrictive procedures. It is defined by the presence of postprandial hypoglycemia (plasma glucose concentration < 55 mg/dL) manifesting with neuroglycopenic symptoms such as confusion or loss of consciousness which resolve when glucose levels are normalized (Whipple’s Triad) (50). PBH is insulin mediated, stimulated by a carbohydrate containing meal, and is distinct from dumping syndrome in that it occurs 1-3 hours after eating without vasomotor symptoms (42).
The reported prevalence of PBH varies widely in the literature depending on the methodology of measurement. In a retrospective nationwide cohort study performed in Sweden, involving >5000 individuals who had undergone bariatric surgery, the rate of hypoglycemia (and related symptoms such as dizziness, visual disturbances, syncope and seizures) as ascertained by diagnosis codes was low but significantly higher in patients without diabetes who had undergone RYGB (0.2%) compared to the general reference population (0.04%) (51). A retrospective cohort study of over 6000 patients who had bariatric surgery at Cleveland Clinic reported a PBH rate of 1.4% (52). A large cross-sectional database analysis of 145,582 US subjects who underwent RYGB and 29,930 who underwent SG showed that only 0.1% and 0.02% had self-reported hypoglycemia as a postoperative complication (53). Another US study involving mailed questionnaires to subjects who had experienced severe or medically confirmed hypoglycemia and the one significant correlate of these severe postoperative hypoglycemic episodes was a history of pre-operative hypoglycemic symptoms (54). The Longitudinal Assessment of Bariatric Surgery 2 study was a U.S. multicenter, prospective study of >2400 participants undergoing bariatric surgery followed for 7 years (55). The rate of symptomatic hypoglycemia at least once during follow up was 39% with 4% reporting continued symptoms over several years, mainly from those who underwent RYGB versus SG or AGB. In this study, higher rates of PBH were seen in people with a history of prediabetes or type 2 diabetes at baseline. A significant risk factor for PBH in this study was the presence of pre-operative hypoglycemia among those with type 2 diabetes. A meta-analysis of studies employing continuous glucose monitoring revealed that the rate of individuals having glucose levels under thresholds ranging from 54-70 mg/dL for some period within 3-7 days was high at approximately 50%. The occurrences of hypoglycemia in these post-surgical individuals happened in the post prandial period as well as overnight (56). Thus, depending on the method of measurement, the range of PBH reported in the scientific literature varies.
The exact pathophysiology of PBH is not entirely understood. In one case series, six individuals with biochemical confirmation of PBH underwent selective arterial calcium stimulation testing followed by partial pancreatectomy (57). Pathological analysis of pancreatic samples confirmed an insulinoma in one, while five had evidence for beta cell hyperplasia and hypertrophy compared to obese controls who had undergone pancreatectomy for pancreatic cancer. The authors of a subsequent study using the same pathology samples taken from the affected post-RYGB patients but compared to otherwise healthy lean and obese controls found no evidence for post-RYGB islet hypertrophy or “nesidioblastosis” and postulated that hyperinsulinemia may instead be due to hyper functioning of existing beta cells (58). A commonly proposed mechanism for such beta cell “hyperfunction” is the large increase in GLP-1 response to meals that occurs after gastric bypass (59-61). In two separate studies, individuals with PBH had higher levels of GLP-1 were generated in response to a mixed meal challenge compared to bariatric patients without symptoms (59,60). However, similar symptoms and effects have not been reported with long-term use of GLP-1 agonists used for the management of type 2 diabetes and obesity. Interestingly, despite large increases in GLP-1 secretion, post-prandial glucagon levels are not suppressed in both non-symptomatic patients after RYGB and PBH patients, nor does glucagon treatment readily reverse this condition.
An alternative explanation for the cause of post-prandial hyperinsulinemic-hypoglycemia after RYGB is a mismatch between the clearance of glucose and insulin after the meal. Gastric emptying is accelerated after RYGB leading to earlier and higher peaks of both glucose and insulin compared to non-surgical controls. Without a pyloric valve regulating nutrient entry to the small intestine, however, glucose levels also fall quickly. Since insulin clearance occurs at a fixed rate, insulin levels may not be able to fall commensurate with the drop in glucose levels, and without a pyloric valve to provide a more gradual entry of food from the stomach to small intestine, a mismatch may ensue.
If suspected, a careful history of symptoms consistent with PBH should be ascertained and other etiologies of hypoglycemia should be ruled out (e.g., medication-induced hypoglycemia and rarely an insulinoma can be unmasked when insulin resistance improves after surgically induced weight loss). Although there is no standardized test to confirm PBH, a mixed-meal tolerance test with confirmatory serum glucose levels both before and at 30-minute intervals after a mixed meal is commonly used (62). Alternatively, continuous glucose monitoring performed in the context of an individual’s normal eating pattern has been demonstrated to be sensitive in detecting PBH (63). Oral glucose tolerance testing is less useful as individuals who have undergone RYGB commonly experience low glucose levels following an oral glucose load without symptoms of hypoglycemia (64,65).
Treatments for PBH ranging from dietary modification to more extreme measures such as gastric bypass reversal have been suggested (66). Recommended dietary modifications consist of small frequent meals that do not result in large, rapid carbohydrate delivery to the small intestine. These meals should be high in fiber and protein and very low in simple carbohydrates (67). Successful use of medications such as nifedipine, somatostatin, and diazoxide (all designed to impair insulin secretion by beta cells) and acarbose (designed to reduce glucose availability in the intestine), have been described in case reports and small series (68-71). Newer medications such as GLP-1 analogs (to prevent endogenous incretin release) (72) and SGLT-2 inhibitors (to reduce blood glucose rises) have been case-reported to work in some individuals (73). As a last resort, symptoms have been shown to resolve with re-introduction of nutrient flow through the stomach and duodenum either by gastric-tube feedings or reversal of the gastric bypass. Due to future risk of diabetes and frequent symptom recurrence, PBH treatment involving distal pancreatectomy is no longer recommended (62).
Cholelithiasis
Rapid weight loss after bariatric surgery promotes gallstone formation by increasing the lithogenicity of bile, with hypersaturation of the bile with cholesterol and with increased mucin production (74,75). Gallbladder hypomotility contributes to this process (76). Further, additional risk factors for cholelithiasis, including obesity, female sex, and premenopausal status, are already prevalent in the bariatric surgery patient population. Indeed, after RYGB, reported incidence of cholelithiasis ranges from 7% to 53%, with most figures around 30%, substantially higher than in the general population (77). A study of patients undergoing SG documented a similarly elevated incidence of radiographic cholelithiasis (78).
Ursodeoxycholic acid, commonly known as ursodiol, can successfully reduce risk of postoperative cholelithiasis. In a multicenter RCT of RYGB patients, ursodiol at any of 3 doses decreased risk compared to placebo, with 43% of patients in the placebo group forming gallstones on ultrasound by the 6-month postoperative time point, vs. 8% of patients in a 300 mg twice daily group. The efficacy of prophylactic ursodiol after bariatric surgery was subsequently confirmed in a meta-analysis of this and 4 other RCTs (79), and an RCT demonstrated that ursodiol decreased cholelithiasis incidence 6 months after SG (78). As a result of these data, oral administration of ursodiol is recommended at 300 mg twice daily after RYGB or BPD/DS and 500 mg once daily after SG (24), although there is not consensus about dose and duration (80). There are emerging data in support of a role of probiotics in prevention of cholelithiasis (80).
Cholecystectomy is sometimes performed at the time of bariatric surgery, but in whom it should be performed is controversial and variable between surgeons (77). Some surgeons perform prophylactic cholecystectomy at the time of surgery; some perform cholecystectomy if preoperative ultrasound reveals gallstones, even if asymptomatic; and some perform concomitant cholecystectomy only if both pathology and symptoms exist.
Nephrolithiasis
Bariatric surgery increases the risk for new-onset nephrolithiasis. This increased risk is procedure-specific and is proportionate to the degree of procedure-induced malabsorption: greatest after BPD/DS, moderate following RYGB, and risk similar to the nonsurgical population following SG and AGB (81-83). For example, in one retrospective cohort study, the comorbidity-adjusted relative hazard of nephrolithiasis was 4.15 (2.16-8.00) after the most malabsorptive procedures and 2.13 (1.30-3.49) after RYGB; the risk after SG and AGB was similar to that of obese controls (82).
The pathophysiologic mechanisms of kidney stone formation after RYGB and BPD/DS include low urinary volume and low urinary citrate, but the driving mechanism relates to high urinary oxalate in the setting of malabsorption (enteric hyperoxaluria) (81,84,85). Normally, dietary calcium binds dietary oxalate, precipitates out as calcium oxalate, and is excreted in the feces. In the setting of malabsorption, non-absorbed fatty acids preferentially bind calcium in the intestine, leaving high concentrations of unbound oxalate that can passively diffuse into the blood, where it is filtered and excreted by the kidneys. Under predisposing conditions—such as low urinary volume—urinary oxalate may precipitate with urinary calcium to form kidney stones. Further, colonic permeability to oxalate may increase with exposure to unconjugated bile salts and long chain fatty acids, both of which increase after bariatric surgery. Finally, it is speculated that postoperative alterations in gut microbiota, and particularly in the oxalate-degrading Oxalobacter formigenes, might also contribute to hyperoxaluria (81,84,85).
Therapeutic strategies to mitigate nephrolithiasis risk after bariatric surgery (Table 7) are similar to those for the general population (86). Fluid intake to achieve a urine volume of at least 2.5 L/day can be a challenge when a small stomach pouch restricts overall intake and a patient has been counseled to drink fluids between rather than with meals. This highlights the need for the sipping of water throughout the day. A registered dietitian can help a patient achieve a diet low in oxalate-rich foods that also meets the patient’s other dietary needs. Some patients may assume that consumption of calcium will increase kidney stone risk and thus may benefit from teaching that adequate calcium consumption (from diet and calcium citrate supplements) is necessary to limit oxalate absorption and avoid enteric hyperoxaluria.
Table 7.
Therapeutic Strategies to Decrease Risk of Kidney Stones
| Strategy | Rationale |
|---|---|
| Hydration to achieve urine volume of ≥ 2.5 L/day | Dilute urine |
| Limitation of oxalate-rich foods (e.g., spinach, nuts, vitamin C) | Limit oxalate absorption |
| Low fat diet | Limit oxalate absorption |
| Adequate calcium consumption (diet ± calcium citrate supplements) | Limit oxalate absorption |
| Low salt and low non-dairy animal protein diet | Increase urinary citrate |
| Potassium citrate therapy if urinary citrate low | Increase urinary citrate |
Bone Loss and Fracture Risk
Bariatric surgery has a significant impact on bone metabolism. All bariatric procedures induce a high postoperative bone turnover state. For example, after RYGB biochemical markers of bone resorption have been shown to double in the first postoperative year (87-90). Bone mineral density (BMD) assessed by dual-energy X-ray absorptiometry (DXA) decreases (87-90), and while there has been concern about potential unreliability of DXA assessment in the setting of marked weight loss and changing soft tissue composition (91,92), declines in BMD have now been demonstrated clearly using quantitative computed tomography (QCT) at the axial skeleton and high-resolution peripheral QCT at the appendicular skeleton (93-98). Decline in BMD has been most consistently reported after RYGB (88,89,99), but also after BPD/DS (100,101) and SG (96,98,102-104). In one recent study comparing cohorts of adults undergoing RYGB vs. SG, some bone density and microstructural parameters changed less after SG than after RYGB, while for other parameters, where was no evidence of smaller effects after SG (98). After AGB, DXA-assessed BMD decreases modestly at the proximal hip but not at the spine (88,89), with reductions in hip density smaller than those after RYGB (105). While some loss of bone mass may be an appropriate physiological response to weight loss, BMD has been shown to decline progressively after RYGB, even after weight stabilization (94,103,106) and mild weight regain (106). Postmenopausal women appear to have the greatest declines in bone density and microstructure after RYGB (107) and after SG (98).
Ultimately, the important question is whether fracture risk increases after bariatric surgery. Studies have now indicated that fracture risk is indeed higher after bariatric surgery in comparison to obese (108-110), non-obese (109), and general population (111) nonsurgical controls. There may be bias introduced when studies identify obese nonsurgical controls based on the assignment of diagnostic codes for morbid obesity, as those nonsurgical patients may be sicker. However, studies with BMI-matching also demonstrate an increase in fracture risk (112,113). Fracture risk after bariatric surgery appears to vary by bariatric procedure, with the risk most clearly defined for RYGB (112). Fracture risk is higher after RYGB than AGB (114,115). Studies published to date about fracture after SG (110,116) have been shorter-term because the procedure is newer; longer-term data are needed before conclusions can be drawn.
Negative skeletal effects resulting from bariatric surgery appear to be multifactorial (90,117-119). Potential mechanisms include the decreased skeletal loading with weight loss; loss of muscle mass; changes in levels of fat-secreted hormones (adipokines), sex steroids, and gut-derived hormones; changes in bone marrow adipose tissue (120); and, importantly, nutritional factors including vitamin D deficiency, inadequate calcium intake, and calcium malabsorption. Intestinal calcium absorption has been shown to decrease after RYGB even in the setting of optimized vitamin D status (99), presumably because the bypassed duodenum and proximal jejunum are usually the predominant sites of active, transcellular, 1,25-dihydroxyvitamin D-mediated calcium uptake, and the distal intestine is unable to compensate. In response to calcium malabsorption after RYGB, parathyroid hormone (PTH) secretion increases, and the effects of PTH include an increase in bone resorption in order to maintain serum calcium concentration. Meanwhile, bone resorption also increases due to non-PTH-mediated processes like mechanical unloading and changes in the hormonal milieu. This mobilization of calcium from the skeleton may actually dampen the need for greater PTH secretion (). A recent study showed that intestinal calcium absorption also decreases after SG, even in the setting of robust vitamin D status, even though it is not a bariatric procedure categorized as malabsorptive (121). Participants with lower postoperative fractional calcium absorption had greater losses of hip BMD, suggesting that calcium malabsorption may contribute to detrimental skeletal effects not only after RYGB but also after SG.

Figure 1.
Effects of RYGB on calcium homeostasis. Reprinted from J Steroid Biochem Mol Biol, Schafer AL, Vitamin D and intestinal calcium transport after bariatric surgery, 173:202-210, 2017 (122), with permission from Elsevier.
Strategies that aim to decrease the risk of postoperative skeletal complications have been included in the AACE/TOS/ASMBS Clinical Practice Guidelines (24) as well as in an additional bone metabolism-focused position statement from the ASMBS (123). A reasonable approach is described in Table 8.
Preoperatively, testing of 25-hydroxyvitamin D level with treatment of vitamin D deficiency is recommended for patients preparing to undergo any bariatric surgical procedure (33). DXA scanning should be performed based on age-appropriate recommendations of the Bone Health and Osteoporosis Foundation (124) or the United States Preventive Services Task Force (125); other patients with risk factors for osteoporosis or fracture, such as postmenopausal women and older men, may be considered for baseline BMD assessment.
Postoperatively, universal supplementation with calcium and vitamin D are necessary after any bariatric surgical procedure, even after procedures without a malabsorptive component; intestinal calcium absorption has been shown to decrease postoperatively not only after RYGB (99) but also after SG (121). Even after AGB, restricted food intake and variety poses a risk for micronutrient deficiencies. After RYGB, SG, and AGB, a total calcium intake of 1200-1500 mg/day from diet and supplements (as needed) is recommended. After BPD/DS, a higher calcium intake may be necessary. Supplemental calcium should be provided as chewable calcium citrate in divided doses. An initial postoperative vitamin D supplement of 3000 IU/day is reasonable for most patients regardless of procedure. Postoperative laboratory monitoring should include 25-hydroxyvitamin D, calcium, albumin, phosphorus, and PTH levels. The vitamin D supplement dose can be titrated to achieve and maintain a 25-hydroxyvitamin D level of at least 30 ng/mL. If secondary hyperparathyroidism is present despite an optimized 25-hydroxyvitamin D level, the most likely cause is inadequate calcium intake or absorption; a low 24-hour urinary calcium level would support this. Increased calcium intake would be appropriate, with follow-up laboratory testing to confirm normalization of PTH level. The PTH level should, of course, be interpreted and targeted based on renal function. Professional organizations have differed in their recommendations about postoperative DXA, in light of the absence of evidence about the utility of such screening. A reasonable approach is to consider DXA after 2 years in those at higher risk for osteoporosis or fracture such as postmenopausal women, older men, and those with other risk factors.
Table 8.
Pre- and Postoperative Skeletal Health Strategies
| Preoperative strategies | |
| Check 25-hydroxyvitamin D and replete low levels | |
| DXA based on age-appropriate screening | |
| Consider DXA in higher-risk individuals such as postmenopausal women, older men, and those with risk factors for osteoporosis or fracture | |
| Postoperative strategies | |
| Supplementation | Calcium, as calcium citrate, to achieve total daily calcium intakes: AGB, SG, RYGB: Calcium 1200-1500 mg/day from diet + supplements BPD/DS: Calcium 1800-2400 mg/day from diet + supplements |
| Vitamin D 3000 IU, titrate to ≥30 ng/mL | |
| Lab monitoring | Calcium, albumin, phosphorus, PTH, 25-hydroxyvitamin D after 3 months, then every 6-12 months |
| 24-hour urinary calcium if additional data is needed (e.g., elevated PTH) | |
| BMD monitoring | DXA based on age-appropriate screening; consider after 2 years in others who are at higher risk such as postmenopausal women, older men, and those with osteoporosis risk factors |
Other strategies which may benefit the skeletal health of the bariatric surgery patient include exercise—particularly weight-bearing and muscle-loading exercise—and higher protein intake, as these mitigate loss of bone mass during non-surgical weight loss in older adults. A randomized controlled trial of a multipronged intervention of exercise, calcium, vitamin D, and protein supplementation was shown to attenuate—although not entirely prevent—postoperative increases in bone turnover markers and declines in BMD after RYGB and sleeve gastrectomy (104).
For those who have had bariatric surgery and are found to be at high risk of fracture, there are very few data to guide the use of antiosteoporosis medications. Antiresorptive agents such as bisphosphonates and denosumab should only be considered after appropriate therapy for calcium and vitamin D insufficiency and confirmation that adequate calcium and vitamin D status are maintained. Otherwise, there is a meaningful risk of medication-induced hypocalcemia (126). If pharmacotherapy is prescribed, a parenterally administered agent is recommended due to concerns about adequate gastrointestinal absorption and potential anastomotic ulceration with orally administered bisphosphonates. Research is needed to guide the use of these medications—either for prevention of bone loss or for treatment of osteoporosis—in the postoperative bariatric surgery population.
WEIGHT REGAIN AFTER BARIATRIC SURGERY
Given that obesity is a chronic disease and sustained weight loss requires ongoing management, understanding the durability of weight loss after bariatric surgery is of critical importance. Unfortunately, published studies reporting weight loss after bariatric surgery thus far tend to be short-term (many with < 5 years follow-up), and longer studies often lack high retention rates and/or adequate control groups (127,128). Additionally, the literature on long-term weight loss mostly addresses AGB and RYGB, and the literature on SG is still emerging. Furthermore, methods of quantifying weight change vary across studies, including percentage excess weight loss (%EWL) and percentage weight loss (%WL) (Table 9), making comparisons between studies challenging. Percentage weight loss (%WL) may be the best method for measuring weight change after bariatric surgery (129), as it is least confounded by preoperative BMI and allows surgical studies to be compared to non-surgical interventions. However, this method is not widely used in the surgical literature.
Table 9.
Hypothetical Comparison of Anthropometrics, including Total Weight Loss, Excess Weight Loss, and Percentage Weight Loss, Following Bariatric Surgery
| Example patient | Baseline | Scenario 1: Post-op BMI 30 kg/m2 |
Scenario 2: Post-op BMI 35 kg/m2 |
|---|---|---|---|
| Weight | 120 kg (264 lbs) | 79 kg (175 lb) | 93 kg (204 lb) |
| Height | 163 cm (64 in) | — | — |
| BMI | 45 kg/m2 | 30 kg/m2 | 35 kg/m2 |
| Ideal Weight (if BMI 25 kg/m2) | 66 kg (145 lbs) | — | — |
| Excess Weight (Weight above ideal weight) |
54 kg (119 lbs) | 14 kg (30 lb) | 27 kg (59 lb) |
| Excess BMI (BMI above 25 kg/m2) |
20 kg/m2 | 5 kg/m2 | 10 kg/m2 |
| Total Weight Loss (baseline weight – post op weight) |
— | 40 kg (89 lbs) | 27 kg (60 lbs) |
| % Weight Loss (Total weight loss/baseline weight x 100) |
— | 44% | 33% |
| % Excess Weight Loss (Total weight loss/excess weight x 100) |
75% | 50% | |
| % Excess BMI Loss (Excess BMI – total BMI loss) |
75% | 50% |
Using RYGB as an example, several studies with long-term follow-up and high retention rates highlight expected weight trajectories ( and Table 10). It is important for patients to understand that the amount of weight loss can be highly variable between people, and that soon after achieving a postoperative weight loss nadir, it is not unusual to have a slight weight regain before achieving a new weight stabilization. These findings were highlighted in an analysis from the Longitudinal Assessment of Bariatric Surgery (LABS) Study, a 10-center observational cohort study in the U.S. that followed 2348 participants after RYGB (n=1738) or AGB (n=610). Serial weight measurements were obtained in person for 82.9% of participants up to 7 years after surgery (10). The weight nadir was typically achieved between 6 months and 2 years after this procedure, with a mean weight loss 7 years after RYGB of 28.4% (95%CI, 27.6-29.2) with 3.9% weight regain having been observed between years 3-7. Grouping individuals by similarly modeled weight-loss trajectories identified six distinct patterns (). Roughly 75% of individuals achieved a 7-year weight loss of 25% or more from baseline (Groups 3 to 6). Less than 5% lost less than 10% of their initial weight while 13.3% lost 45% or more. These patterns of weight loss closely mirrored achieved weight loss by 6 months and all but one group experienced some weight rebound between postoperative years one to six (10).
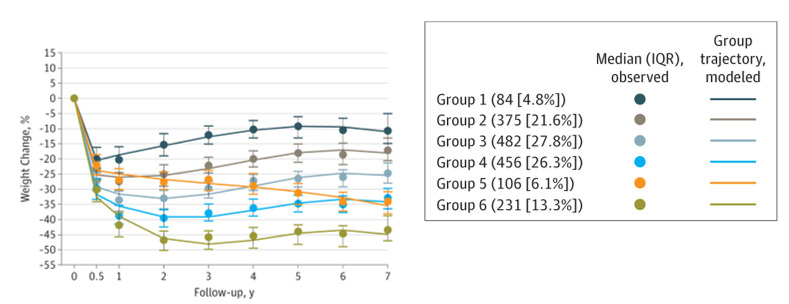
Figure 2.
Weight change trajectory groups following RYGB. Lines indicate modeled group trajectories; data markers and median values; bars, interquartile range (IQR) of observed data. Negative value indicates weight loss from baseline (10).
Adams et al. prospectively followed a cohort of 417 subjects undergoing RYGB at a Utah-based surgical group (130) for 6 years, 92.6% of whom had follow-up weights, mostly obtained via in person measurement or medical chart review. Weight change was compared to 2 control groups: those who sought but did not undergo surgery (72.9% follow-up) and matched controls from a local healthcare database (96.9% follow-up). The RYGB group had the greatest mean adjusted weight loss from baseline to postoperative year 2 at 34.9%, decreasing to 27.7% in postoperative year 6. The authors report the absolute difference between these two figures as “percent weight regain” of 7.2%. Additionally, among RYGB patients, 94% had lost >20% of baseline weight at year 2, though 76% had maintained >20% weight loss at year 6. The control groups experienced negligible weight change.
A Veterans Administration (VA) retrospective cohort study evaluated 10-year weight loss outcomes among 1787 individuals who underwent RYGB, comparing these to 5305 non-surgical matches derived from the VA electronic health record (131). Among eligible patients, 81.9% of RYGB patients and 67.4% of non-surgical matches had follow-up data at 10 years. Percentage weight loss in the RYGB group was 31% (n= 1755) at year 1 and 28% (n=564) at year 10. The control group had lost only modest amounts of weight in follow-up, and the difference in weight loss between RYGB and controls was calculated at 30% and 21% in postoperative years 1 and 10, respectively.
Christou et al. of McGill University retrospectively studied 272 patients who had undergone RYGB, 83% of whom were available for in-person or phone follow-up (132). Among all patients, the greatest %EWL was 89% at the 2.5 years postoperative time-point, and this reduced to 68.1% at the 12 years postoperative time-point. Thus, approximately 18% of excess weight loss in the second year was regained by year 12. At 10 years, among patients with a starting BMI <50 kg/m2, “excellent” surgical response (postoperative BMI <30 kg/m2) and “good” surgical response (postoperative BMI 30-35 kg/m2) were achieved in 51% and 29%, respectively. Among those with a baseline BMI >50 kg/m2, the results were less positive: 13% achieved an excellent response, and 29% achieved a good response. The rates of follow-up were similar between the two groups.
Carbajo et al. of Spain performed a database analysis of 1200 patients who underwent one-anastomosis gastric bypass (a modification of RYGB) and had at least 6 years follow-up (133). Mean preoperative BMI was 46 kg/m2 (range, 33-86 kg/m2). Among the 1200, follow-up rates at 6, 8, 10 and 12 years were 87% (n=233), 74% (n=607), 72% (n=759) and 70% (n=839), with roughly half followed up in person and half via electronic correspondence. %EWL was 77% for 6-year follow-up, 73% for 8-year follow-up, and 70% for 10- and 12-year follow-up. Percentage weight loss in the first 5 years of operation was not reported.
Pories et al. of East Carolina University School of Medicine retrospectively evaluated %EWL in patients who underwent RYGB from 1980-1994 (134). Among the 608 operated on, 574 were alive at the time analysis, and 553 of those remained in contact (i.e., 96% follow-up). Among the 553, 49% were examined in person, and the remainder were interviewed by telephone. Mean %EWL values at years 1, 2, 10, and 14 were 69% (n=506), 58% (n=407), 55% (n=158), and 49% (n=10), respectively. Thus, the average excess weight loss at 14 years is 20% less than at year 1.
Overall, among RYGB studies with high retention rates, the greatest average weight loss (nadir) is typically reported in the first two years with %EWL ranging from 69-89% and %WL of 31-35%. In general, about 10-20% of the maximum weight lost after surgery is regained when patients are followed for six years or longer. However, %EWL remains between 49-70% and %WL nearly 30%, which far exceeds any non-surgical weight loss interventions.
Table 10.
Observational Studies of Long-Term Weight Loss Following RYGB
| Author (Year) Study Type |
Study size (% follow up) | Weight loss at follow-up |
|---|---|---|
| Courcoulas (2018) Prospective |
N=1130 (86%) | 28.4% WL at 7 years |
| Adams (2012) Prospective |
N=417 (92.6%) | 27.7% WL at 6 years |
| Maciejewski (2016) Retrospective |
N=688 (81.9%) | 28% WL at 10 years |
| Christou (2006) Retrospective |
N=288 (83%) | 68.1% EWL at 12 years |
| Carbajo (2017) Retrospective |
N=1200 (87% at 6 years, 74% at 8 years, 72% at 10 years, 70% at 12 years) | 77% EWL at 6 years 73% EWL at 8 years 70% EWL at 10 years 70% EWL at 12 years |
| Pories (1995) Retrospective |
N=574 (96%) | 55% EWL at 10 years |
In the LABS study, 7-year weight loss after AGB averaged only 15% with 25% losing ≤ 5% of baseline weight and another 5% regaining all their lost weight and more (10). Data on weight loss following SG is still emerging, but typically runs roughly 2% to 5% less than RYGB by either %EWL or %TWL criteria, with greater variability between patients (13,135-138).
Medical Management of Postoperative Weight Regain
In observational studies, clinical predictors of insufficient weight loss or weight regain after bariatric surgery have identified specific diet and exercise practices, female sex, older age, higher initial BMI, presence of T2D, psychological factors, and non-white race, although the influence of any individual factor is relatively small (10,139,140). While the actual physiology that explains the long-term weight rebound following both RYGB and SG or why some individuals achieve 50% weight loss (or more) and others regain all their lost weight remains unknown at present, it is possible traditional influencers of body weight are playing a role, including genetics (141,142) and the postoperative use of medications that promote weight gain (143).
Interventions to stabilize or restore weight loss that have compared lifestyle or psychological support to usual care after surgery have shown (with some exceptions) to be minimally effective, but the studies conducted thus far have been relatively small (139). Several observational studies suggest that medical (drug) weight loss therapy may be a promising modality to aid in weight loss after bariatric surgery. The largest published study to date on the use of pharmacologic agents to reverse weight regain or weight loss plateau came from 319 patients who underwent RYGB (n=258) or SG (n=61) at 2 academic centers and had been prescribed one or more weight loss medications (144) with at least 1 year of follow-up (145). The medications included FDA-approved weight-loss drugs (e.g., phentermine, liraglutide, orlistat) and off-label use of medications with potential weight-lowering effects (e.g., topiramate monotherapy, metformin, pramlinitide, and canagliflozin). The medications were more often started for weight regain (78.5%) than weight loss plateau (21.5%), and the mean start time of a medication was earlier after SG (mean of 23.2 months) than RYGB (mean of 59.3 months). Overall, 54% of patients lost at least 5% of weight, 30% lost at least 10%, and 15% lost more than 15%. Topiramate use was associated with the highest success, with a 1.9 odds ratio of achieving at least 10% weight loss. Other small observational studies have shown efficacy of topiramate, liraglutide, phentermine, and phentermine/topiramate combination for post bariatric surgery weight loss (146-150). There have been several trials supporting the efficacy of GLP-1 analogs liraglutide and semaglutide to treat postoperative weight regain (151,152).
While historically weight loss variability or regain after weight-loss surgery has been attributed to “poor habits” or “failure” on the patient’s part to adhere to recommended food intake, it is now recognized that such variability is similar to other chronic diseases where some individuals respond well to certain therapies while others do not or in which progression of the underlying disease state necessitates combination therapies (such as in T2DM as the islet cell impairment progresses over time). It is therefore important to continue to support the patient who experiences postoperative weight regain by emphasizing continued healthy lifestyle practices, identifying medical conditions or medications that might be contributing to their weight gain and either stopping them or switching to weight neutral medications, and considering adding in weight loss medications (153).
link

