Donor characteristics
Skeletal muscle biopsies of the vastus lateralis were obtained from 6 RA patients, aged 61–79 (BMI 21.2–31.9) and 5 age-matched healthy controls, aged 62–76 (BMI 20.6–32.9). All 6 RA patients were female (Supplementary Table 1). Of the healthy controls, 3 were female and 2 were male. RA donors, for which the DAS28 score was known, had disease scores indicating moderate disease activity (3.2 < DAS28 ≤ 5.1). Other demographic characteristics and laboratory values of the RA patients and controls are shown in Supplementary Table 1.
Contractile forces and kinetics of RA and control myobundles treated with IFN-γ with and without IL-6
To identify cytokines that differentially affected RA and healthy control myobundle contractile force, preliminary experiments were first conducted with RA myobundles from one donor which were differentiated for 7 days and exposed to combinations of IL-1β, IL-6, TNF-α, and IFN-γ for 3 days (Supplementary Fig. 1A, B). At the dose used (5 ng/mL), only IFN-γ caused a reduction in specific twitch or tetanus force.
Next, we compared the response of 5 RA donors and 3 healthy control donors that were differentiated for 7 days and then treated for 3 days with either no cytokines, 5 ng/mL IFN-γ, 5 ng/mL IL-1β + 5 ng/mL IL-6 + 5 ng/mL TNF-α, or 5 ng/mL IL-1β + 5 ng/mL IL-6 + 5 ng/mL TNF-α + 5 ng/mL IFN-γ, followed immediately by measurement of contractile force and determination of specific contractile force (force per cross-sectional area) (Supplementary Fig. 2). None of the treatments affected contractile force in healthy myobundles. Treatment with 5 ng/mL IFN-γ significantly reduced all contractile forces in RA but addition of IL-1β, TNF-α, and IL-6 did not further reduce contractile force.
Based on this we examined the RA and control myobundle response after 7 days treatment to IFN-γ and IL-6. While we did not observe an effect of IL-6 after 3 day treatment, we continued to use it because of its role in RA. RA and age-matched control myobundles were differentiated for 7 days to allow myobundles to mature and then treated chronically with either 5 ng/mL IFN-γ, 5 ng/mL IL-6, or 5 ng/mL IFN-γ + 5 ng/mL IL-6 for 7 days, from day 7 to day 14 of differentiation, followed by testing for contractile force, histology, protein expression, and RNA sequencing.
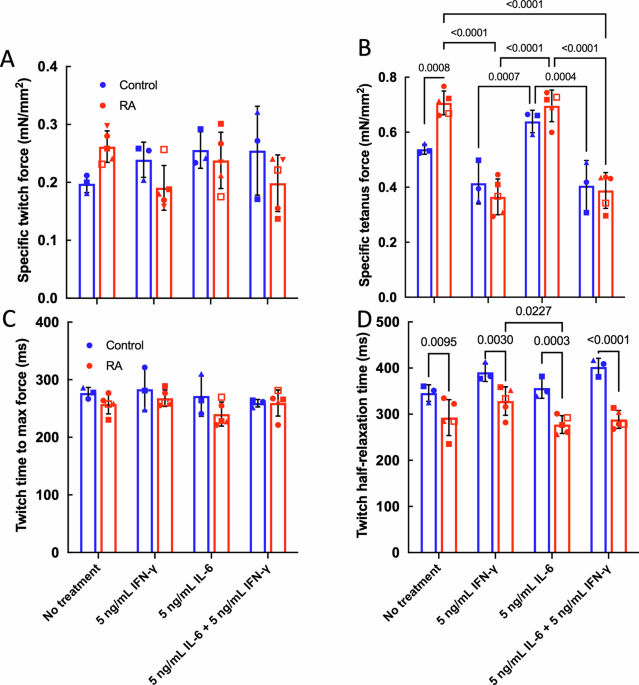
A two-way ANOVA indicated that the interaction between RA disease and cytokine treatment significantly affected specific twitch force (p = 0.0484), but RA and cytokine treatment alone was not significant (Fig. 1A). A post hoc Tukey’s test did not detect any pairwise differences. A two-way ANOVA indicated that cytokine treatment (p < 0.0001) and the interaction between cytokine treatment and RA disease (p = 0.0087) significantly affected specific tetanus force. A post-hoc Tukey test indicated that without any treatment, RA myobundles had higher specific tetanus forces than control myobundles (Fig. 1B), as we noted previously24. In RA myobundles, treatment with IFN-γ as well as IFN-γ plus IL-6 reduced specific tetanus force, whereas for control myobundles the addition of IL-6 led to greater tetanus forces relative to control myobundles treated with IFN-γ alone. Based on these results, IL-6 appeared to have little effect on RA myobundle force production for the conditions studied.
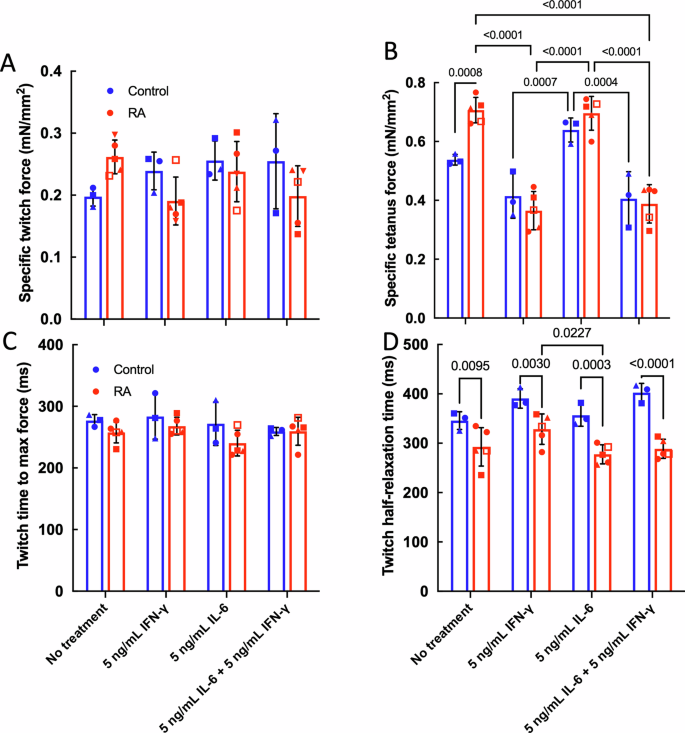
From a two-way ANOVA, RA disease significantly affected twitch time to maximum force (p = 0.0107) but no significant difference was observed with a post hoc Tukey test (Fig. 1C). For twitch half-relaxation time, RA myobundles were significantly different than control myobundles for all treatment conditions (Fig. 1D).
The trends in contractile force observed for the pooled data in response to cytokine treatment were also observed with the individual donors. For control donors, there was no decrease in specific twitch force with either IFN-γ or IL-6 alone or together (Supplementary Fig. 3), although IL-6 did lead to an increased twitch force with C109 (Supplementary Fig. 3). For specific tetanus force. only one control donor (C111) showed a decrease in contractile force with IFN-γ treatment. Two RA donors exhibited a decrease in twitch force following treatment with IFN-γ treatment, RA104 and RA107 (Supplementary Fig. 4). All RA donors showed a decrease in specific tetanus force after IFN-γ treatment, and 3 donors showed decreased specific tetanus force with IFN-γ and IL-6 treatment relative to IL-6 treatment (Supplementary Fig. 4).
In control myobundles, IFN-γ with and without IL-6 or IL-6 alone reduced fatigue relative to no treatment (Supplementary Fig. 5). In RA myobundles, fatigue was unaffected by IFN-γ or IL-6 treatment.
Myofiber analysis of RA and control myobundles treated with IFN-γ with and without IL-6
Longitudinal sections of cytokine-treated myobundles immunostained for sarcomeric α-actinin (SAA) and nuclei were examined between RA donors and control donors (Fig. 2A, B) and appeared similar under all conditions. There was no effect of RA disease or cytokine treatment on SAA+ area (Supplementary Fig. 6A), myofiber orientation (Supplementary Fig. 6C), and fiber length (Supplementary Fig. 6D). A two-way ANOVA indicated that RA disease significantly affected fraction nuclei in SAA+ fibers (p = 0.0196) (Supplementary Fig. 6B) and percent striated fibers (p < 0.0001) (Supplementary Fig. 6E). Surprisingly, in RA myobundles, IFN-γ treatment led to a higher fraction of nuclei in SAA+ striated fibers relative to healthy myobundles (Supplementary Fig. 6B). The percent of striated fibers was larger in RA myobundles than control myobundles for the cases of no treatment, IL-6, and IFN-γ with IL-6 (Supplementary Fig. 6E).
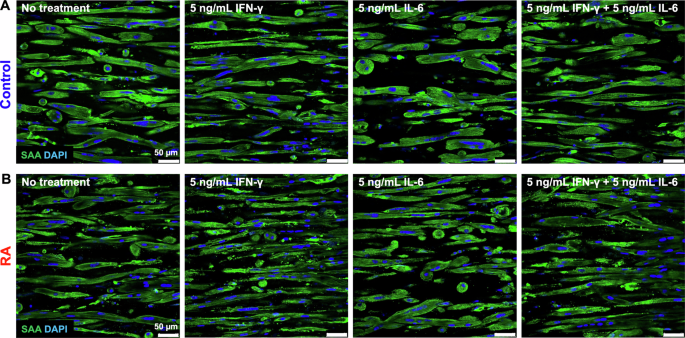
A Control and (B) RA myobundles were fixed and immunostained for SAA and nuclei at day 14 of differentiation after 7 days of treatment with IFN-γ with and without IL-6. Results are shown for RA donor RA106 and control donor C111. Scale bar = 50 µm.
Muscle protein analysis of RA and control myobundles treated with IFN-γ with and without IL-6
Western blot for myosin heavy chain (MYH), soluble IL-6 receptor (IL-6R), and myosin light chain (MYL) were compared to a β-actin control for 5 RA donors and 3 controls (Fig. 3A) and the results quantified (Fig. 3B–D). Since the IL-6R signal may have been affected by the β-actin band, particularly for RA myobundles, we ran separate gels in which we stained for IL-6R and β-actin on different lanes of the gel (Supplementary Fig. 7). As a result, background was reduced and the effect on IL-6R of treatment of RA myobundles with IFN-γ with or without IL-6 is noticeable.
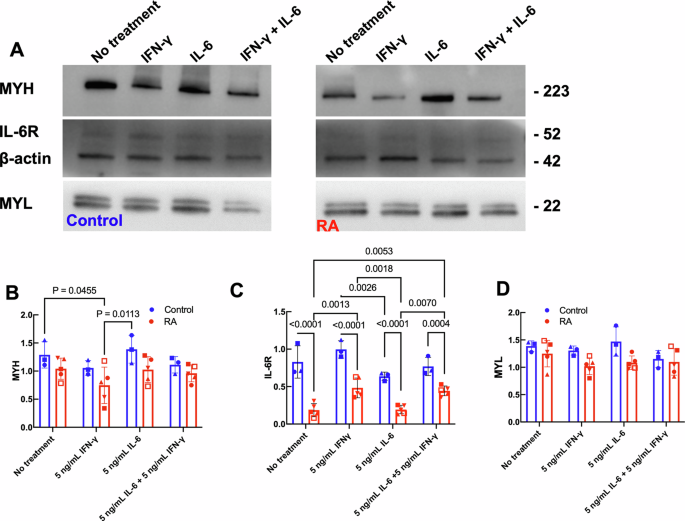
A Western blot of MYH, IL-6R, and MYL in one RA donor (RA106) and one control (C109) on day 14 of differentiation after 7 days of treatment with IFN-γ with and without IL-6. Quantified expression of (B) MYH, (C) IL-6R, and (D) MYL normalized to β-actin for RA and controls. N = 5 RA donors (RA104 ( ), RA106 (
), RA106 ( ), RA107(
), RA107( ), RA108 (
), RA108 ( ), and RA109(
), and RA109( )), 3 control donors (C109 (
)), 3 control donors (C109 ( ), C111(
), C111( ), C113(
), C113( )). 4 myobundles were pooled together per condition per donor to obtain protein sample. Data are represented as mean ± S.D. and analyzed with two-way ANOVA, followed by Tukey HSD post-hoc.
)). 4 myobundles were pooled together per condition per donor to obtain protein sample. Data are represented as mean ± S.D. and analyzed with two-way ANOVA, followed by Tukey HSD post-hoc.
A two-way ANOVA indicated that in RA myobundles there was no effect of disease type or cytokine treatment (IFN-γ with or without IL-6) on MYH protein levels. There was an interaction effect between the disease and cytokine treatment conditions (p = 0.0455) as noted by the differences between RA myobundle treated with IFN-γ and control bundles that were untreated or treated with IL-6 (Fig. 3B). Additionally, RA (p < 0.0001) and cytokine treatment (p < 0.01) significantly affected IL-6R levels (Fig. 3C). Furthermore, a significant interaction effect was observed between disease and treatment conditions (p = 0.0004) (Fig. 3C). RA disease and cytokine did not affect MYL levels (Fig. 3D).
IL-6R levels were lower in RA myobundles compared to controls for IFN-γ treatment (Fig. 3C). Treatment of control myobundles with IFN-γ with or without IL-6 elevated IL-6R, whereas only the combination of IFN-γ and IL-6 affected IL-6R levels in RA myobundles. This finding is consistent with force results where IL-6-treated RA myobundles produced greater specific tetanus force compared to IFN-γ-treated RA myobundles (Fig. 1B). These results suggest an inverse association between RA force production and RA contractile protein levels in response to IL-6, which is supported by linear regression of specific tetanus force v. IL-6R levels (Supplementary Fig. 8). No correlation between specific tetanus force and protein levels was observed for MYH or MYL. These results suggest additional factors beyond MYL and MYH contractile protein levels affected the response of RA myobundles to IFN-γ.
RNA-sequencing analysis of RA and control myobundles treated with IFN-γ with and without IL-6
Principal components analysis and heatmaps
To determine genes and pathways most affected in RA versus control myobundles treated with IFN-γ with and without IL-6 for 7 days, RNA-sequencing was performed for 3 RA donors and 3 age-matched controls after cytokine treatment. A principal components analysis (PCA) plot of the gene expression results indicated that IFN-γ produced the largest effect on PC1 for RA and control myobundles. Samples that were either untreated or treated with IL-6 appeared to cluster together, whereas samples treated with IFN-γ with or without IL-6 clustered together (Fig. 4A). IL-6 had minimal effect in PC1 or PC2 (Fig. 4A). These trends agreed with our force results for RA myobundles in which IFN-γ reduced specific tetanus force whereas IL-6 had no effect on force alone or when combined with IFN-γ (Fig. 1A, B). PC2 within the PCA plot showed variability among donors, in which control donors were more variable than RA donors with or without IFN-γ treatment (Fig. 4A). However, it should be noted that the 8% variance associated with PC2 was significantly less than the 60% variance associated with PC1 (Fig. 4A).
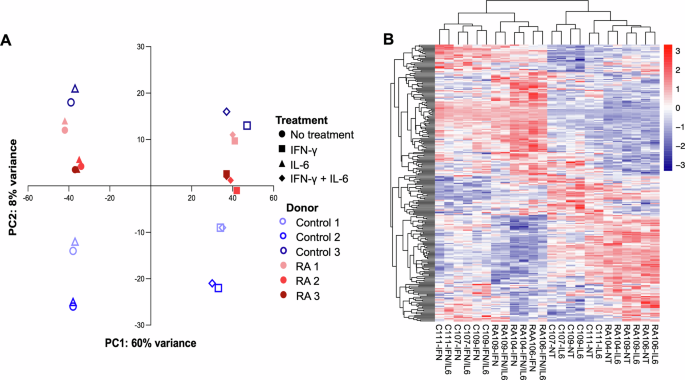
A Principal components analysis (PCA) and (B) heatmap with dendrograms of gene expression of RA and control myobundles treated with IFN-γ with and without IL-6 for 7 days relative to corresponding untreated conditions (NT). RA and control myobundles were flash frozen on day 14 of differentiation prior to RNA isolation. N = 3 RA donors (RA104, RA106, RA109 corresponding to RA 1, RA 2 and RA 3, respectively), 3 control donors (C107, C109, C111, corresponding to Control 1, Control 2 and Control 3, respectively); n = 4 myobundle RNA samples per donor, except n = 3 myobundles for C111 IFN-γ condition. Myobundles were pooled together per condition per donor.
A heatmap and dendrogram were produced for the top 250 differentially expressed genes for cytokine-treated control myobundles and RA myobundles relative to untreated conditions (Fig. 4B). Each column represents the results for a separate donor. The dendrogram indicates that the primary clustering of the donor samples is between those with and without IFN-γ treatment. Within each of those conditions, the control and RA samples form separate clusters. For each donor, IL-6 treatment is adjacent to the corresponding untreated or IFN-γ treatment case, which is consistent with the force results in which treatment with IL-6 did not affect contractile force.
Differential changes in gene expression after IFNγ treatment
Since treatment with IFN-γ alone induced the largest effect on gene expression for RA myobundles and controls (Fig. 4), we focused our attention on that treatment group. Gene expression was significantly affected in 573 genes for RA and 545 different genes for controls for FDR ≤ 0.05 and |log2FC| > 2 (Fig. 5A). Gene expression was significantly affected in RA and controls for 421 of the same genes (Fig. 5A). Volcano plots displaying gene expression results for treatment with IFN-γ in control and RA myobundles are shown in Fig. 5B, C, respectively. The top 10 most significantly upregulated genes that also pertain to one or more gene sets that were enriched in RA and controls, respectively, are labeled in the volcano plots. The trends are very similar to those observed for the conditions that included IL-6 treatments (Supplementary Fig. 9).
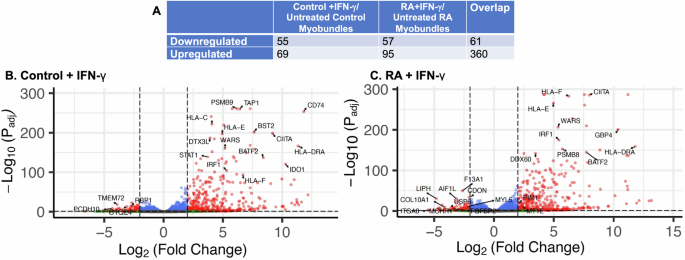
RA and control myobundles were treated with IFN-γ for 7 days from day 7 to day 14 of differentiation. A Number of significant genes between IFN-γ-treated RA myobundles and IFN-γ-treated controls, (B) volcano plot showing up- and down-regulated genes for IFN-γ-treated controls relative to untreated controls, and (C) volcano plot showing up- and down-regulated genes for IFN-γ-treated RA myobundles relative to untreated RA myobundles. Gene expression results for IFN-γ-treated RA and control myobundles were normalized to respective untreated controls. N = 3 RA donors, 3 control donors; n = 4 myobundle RNA samples per donor, except n = 3 for RA111 IFN-γ condition. The donors are the same as those listed in Fig. 4. Myobundles were pooled together per condition per donor. |log2FC| ≥ 2, padj < 0.05.
Although IFN-γ treatment did not reduce MYH and MYL contractile protein levels in RA myobundles compared to controls, the antibodies were not specific for individual isoforms. To assess any effect on the expression of any effect on contractile protein isoforms, we examined expression at the gene level. Among these, MYL5 encodes one of the regulatory myosin light chains, a component of the motor protein myosin. MYL5 was significantly downregulated in IFN-γ-treated RA myobundles (Log2FC = −2.316, Padj = 1.98 × 10−12) and to a lesser extent in controls (Log2FC = −1.559, Padj = 5.85 × 10−8) (Fig. 5C). Genes with |Log2FC| > 1 and padj < 0.05 and are expressed in either IFN-γ treated RA or control myobundles relative to their corresponding untreated conditions are listed in Supplementary Data 1.
Although the primary focus of the RNA sequencing was to examine genes affected after treatment with IFN-γ, we also directly compared differential gene expression of RA myobundles to control myobundles (Supplementary Fig. 10). Since fewer genes were differentially expressed, we set a threshold of |Log2FC| > 1 and padj < 0.05. Five of the differentially expressed genes were also differentially expressed in INF-γ treated RA (AEBP1, LBP, GREM1, lncMYOD1) or control (HMCN1) myobundles relative to their corresponding untreated case (Fig. 5A and Supplementary Data 1).
Gene set enrichment analysis
GSEA was performed using the Hallmark gene set database to compare expression datasets between RA myobundles and controls with and without IFN-γ treatment. When comparing untreated RA myobundles with untreated control myobundles, enrichment results for RA showed that of the 13 out of 50 gene sets that were upregulated, 2 gene sets—oxidative phosphorylation and MYC targets v1—were significantly enriched with an FDR < 0.05 and nominal p value < 0.05 (Table 1). For untreated controls, epithelial mesenchymal transition was the only gene set upregulated relative to untreated RA myobundles with an FDR < 0.05 and nominal p value < 0.05 (Table 1).
Compared to control myobundles treated with IFN-γ, IFN-γ-treated RA myobundles showed no significant gene set enrichment. IFN-γ-treated control myobundles showed 1 gene set that was significantly enriched: epithelial-mesenchymal transition, as was observed without IFN-γ treatment (Table 1).
Next, gene sets enriched in control myobundles treated with IFN-γ were compared to untreated control myobundles. Compared to untreated controls, IFN-γ-treated controls presented 13 statistically significant gene sets involving IFN-γ response, IFN-α response, inflammatory response, IL-6-JAK/STAT3 signaling, TNF-α signaling, apoptosis, and IL-2-JAK/STAT5 signaling (FDR < 0.05, nominal p value < 0.05) (Table 2). Untreated controls showed 1 gene set that was significantly enriched: Hallmark angiogenesis at FDR = 0.05, nominal p value < 0.05 (Table 2).
Enrichment results comparing IFN-γ-treated RA myobundles with untreated RA myobundles showed 16 statistically significant gene sets involving IFN-γ response, IFN-α response, inflammatory response, IL-6-JAK/STAT3 signaling, TNF-α signaling, apoptosis, IL2-JAK/STAT5 signaling, hypoxia, and unfolded protein response (FDR < 0.05, nominal p value < 0.05) (Table 2). Untreated RA myobundles showed no significant results. Interestingly, gene sets for hypoxia, mTOR1 signaling, and the unfolded protein response were enriched in IFN-γ-treated RA myobundles, but not IFN-γ-treated control myobundles (Table 2).
Within the gene set involving genes upregulated in response to hypoxia, only 2 genes were significantly upregulated (|log2FC| ≥ 2, padj < 0.05) in IFN-γ-treated RA myobundles but not IFN-γ-treated controls: PIM1 (log2FC = 2.078; padj = 1.17 × 10−18) and MT-1E, an isoform of MT-1 (log2FC = 2.359; padj = 1.78 × 10−12). Within the gene set involving genes upregulated for the MTOR1 response and the unfolded protein response, no genes were significantly upregulated in IFN-γ-treated RA myobundles or IFN-γ-treated controls.
Contractile forces and kinetics of IFN-γ-treated RA myobundles after treatment with methotrexate and tofacitinib
We next investigated whether specific therapeutics, clinically used for RA treatment, can recover RA myobundle function during continued exposure to IFN-γ after a functional deficit was produced. Given that IFN-γ targets the JAK/STAT pathway, we treated IFN-γ-treated RA myobundles with clinically relevant doses of the JAK-inhibitor tofacitinib (TOF)27,28 or methotrexate (MTX)29,30, the leading DMARD for RA therapy. RA myobundles were treated with 5 ng/mL IFN-γ for 7 days, from day 7 to day 14 of differentiation. After 4 days of exposure to IFN-γ, myobundles were treated with either 500 nM MTX, DMSO vehicle control for MTX, or 1 µM TOF for 3 days while also treated with IFN-γ. Endpoint testing occurred on day 14 of differentiation.
In RA myobundles, a one-way ANOVA indicated that treatment significantly affected specific twitch force (p < 0.01) (Fig. 6A) and specific tetanus force (p < 0.0001) (Fig. 6B). As expected, IFN-γ reduced specific twitch force (Fig. 6A) and specific tetanus force (Fig. 6B) in RA myobundles. In IFN-γ-treated RA myobundles, treatment with TOF fully recovered specific twitch force (Fig. 6A) and specific tetanus force (Fig. 6B). Longitudinal sections of myobundles treated with IFN-γ with or without TOF were immunostained for SAA and nuclei for one RA donor. The morphology of RA myobundles treated with TOF appeared comparable to untreated controls (Supplementary Fig. 11). MTX did not show a recovery in specific twitch or tetanus force when the results for 3 RA donors were averaged together (Fig. 6A, B).
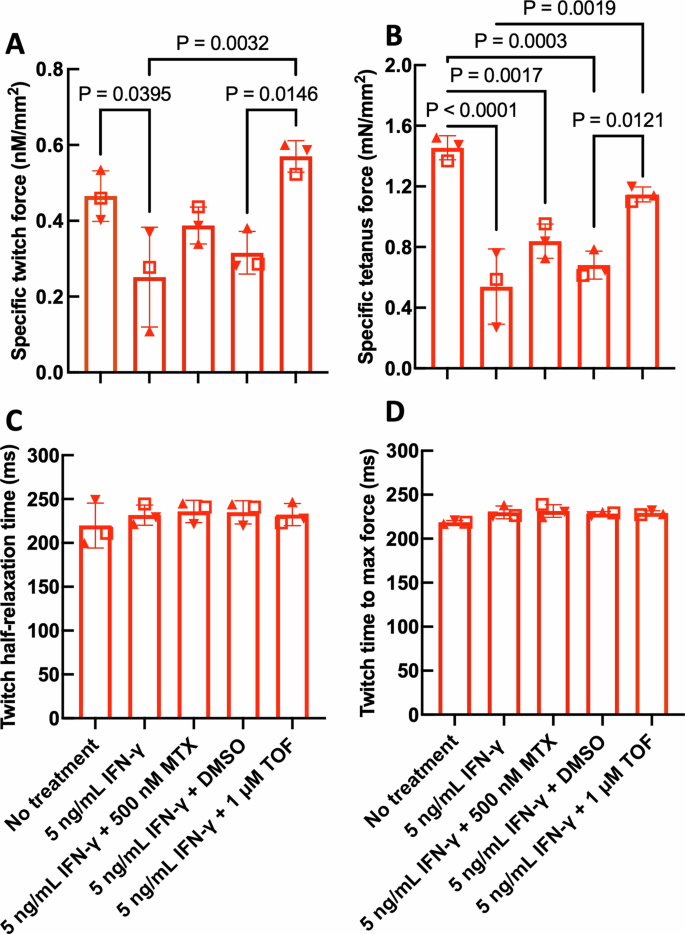
A Specific twitch force, (B) specific tetanus force, (C) twitch time to max force, and (D) twitch half-relaxation time of RA myobundles treated with IFN-γ for 7 days with or without MTX or TOF for 3 days, and force tested on day 14 of differentiation. N = 3 RA donors (RA107 ( ), RA108 (
), RA108 ( ), RA109(
), RA109( )); twitch force and kinetics, n = 4–7 myobundles; tetanus force, n = 3–7 myobundles. Data are represented as mean ± S.D. and analyzed one-way ANOVA followed by Tukey HSD post-hoc test.
)); twitch force and kinetics, n = 4–7 myobundles; tetanus force, n = 3–7 myobundles. Data are represented as mean ± S.D. and analyzed one-way ANOVA followed by Tukey HSD post-hoc test.
Individual assessment of donors shows that the twitch force response was variable with 2 of 3 donors showing a response to IFN-γ (Supplementary Fig. 12A–C). For those two donors (RA108 and RA109), both showed an improvement with TOF while only RA108 showed an improvement with MTX. For tetanus, all donors responded to TOF and none of 3 RA donors exhibited full recovery of force production in response to MTX (Supplementary Fig. 12D–F).
Muscle protein analysis of IFN-γ-treated RA myobundles after tofacitinib treatment
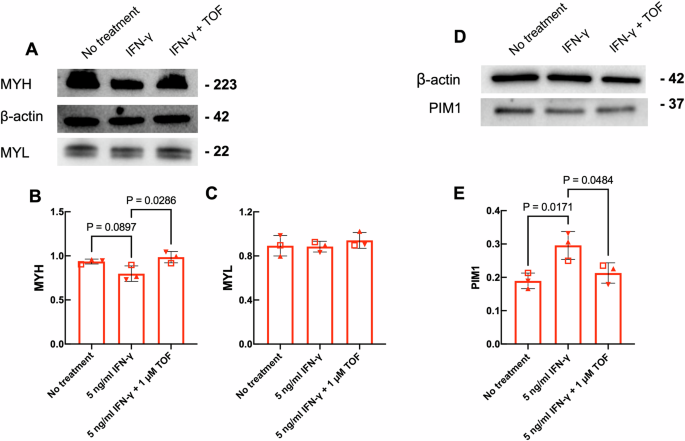
Western blot for MYH and MYL were compared to a β-actin control for 3 RA donors. In RA myobundles compared to untreated controls, IFN-γ treatment showed a trend towards reduced but not significant MYH levels in RA myobundles, which increased significantly after treatment with TOF (Fig. 7A, B). MYL levels in RA myobundles were unaffected by IFN-γ treatment with or without TOF (Fig. 7A, C).
A Western blot of MYH and MYL in one RA donor on day 14 of differentiation after 7 days of IFN-γ treatment with or without TOF for 3 days. Quantified expression of (B) MYH and (C) MYL normalized to β-actin. N = 3 RA donors (RA107 ( ), RA108 (
), RA108 ( ), RA109 (
), RA109 ( )). 4 myobundles were pooled together per condition per donor to obtain protein sample. Data are represented as mean ± S.D. and analyzed using one-way ANOVA, followed by Tukey HSD post-hoc test. D Western blot of PIM1 protein in IFN-γ-treated RA myobundles after treatment with tofacitinib. E Quantified expression of PIM1 normalized to β-actin.
)). 4 myobundles were pooled together per condition per donor to obtain protein sample. Data are represented as mean ± S.D. and analyzed using one-way ANOVA, followed by Tukey HSD post-hoc test. D Western blot of PIM1 protein in IFN-γ-treated RA myobundles after treatment with tofacitinib. E Quantified expression of PIM1 normalized to β-actin.
PIM1 protein expression after treatment of RA myobundles with IFN-γ with and without tofacitinib
A consistent finding from RNA sequencing was elevation of PIM1 and MT-1E after IFN-γ treatment in RA myobundles but not control myobundles. To assess whether this led to altered protein expression which could be restored by tofacitinib treatment, Western blots for PIM1 were performed with the same samples used to measure MYH and MYL (Fig. 7D). Quantification of band intensities indicated that PIM1 levels increased after treatment with 5 ng/mL IFN-γ for 7 days but did not change if 1 µM tofacitinib was added with the IFN-γ (Fig. 7E). We did not observe expression of MT-1E by Western blot.
Inflammatory marker expression in IFN-γ-treated RA myobundles after treatment with methotrexate and tofacitinib
To examine how inflammatory factors relevant to RA pathology were influenced in RA myobundles by exposure to IFN-γ with and without treatment with MTX or TOF, we assessed expression of genes IFN-γ, IL-1β, IL-6, and TNF-α for 3 RA donors. Although not statistically significant, there was a trend for upregulated IFN-γ and IL-1β expression in response to IFN-γ treatment, and a trend for downregulated IFN-γ and IL-1β expression when treated with TOF (Fig. 8A, B). IFN-γ significantly upregulated IL-6 expression in RA myobundles (Fig. 8C). MTX and TOF each restored IL-6 expression levels to baseline (Fig. 8C). Although not statistically significant, TNF-α showed a trend of upregulated expression with IFN-γ treatment (Fig. 8D). TOF downregulated TNF-α expression to levels comparable to untreated controls (Fig. 8D).
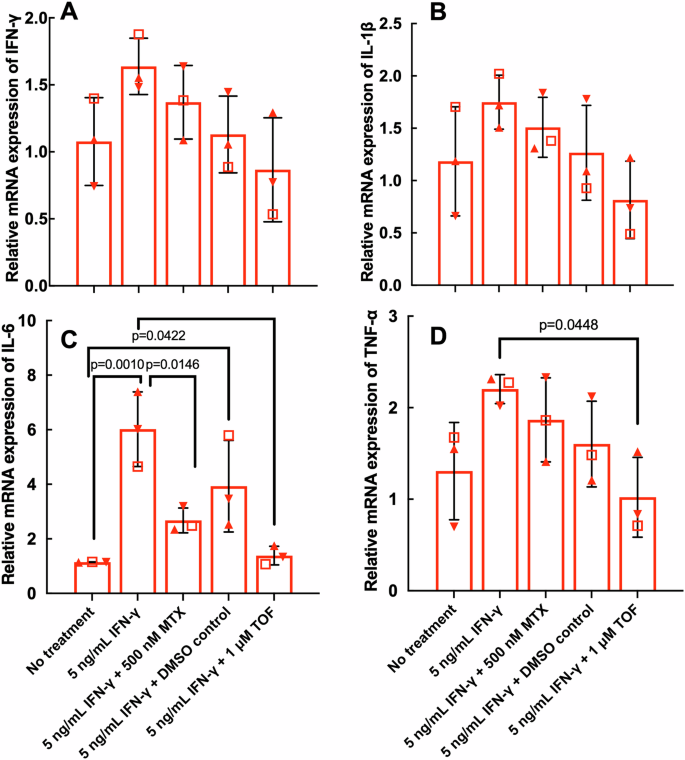
RA myobundles were treated with IFN-γ for 7 days with or without MTX or TOF for 3 days, and flash frozen on day 14 of differentiation. Quantified gene expression of (A) IFN-γ, (B) IL-1β, (C) IL-6, and (D) TNF-α normalized to GAPDH. N = 3 RA donors (RA107 ( ), RA108 (
), RA108 ( ), RA109 (
), RA109 ( ), n = 3 myobundles per donor. Data are represented as mean ± S.D. and analyzed using one-way ANOVA, followed by Tukey HSD post-hoc test.
), n = 3 myobundles per donor. Data are represented as mean ± S.D. and analyzed using one-way ANOVA, followed by Tukey HSD post-hoc test.
link