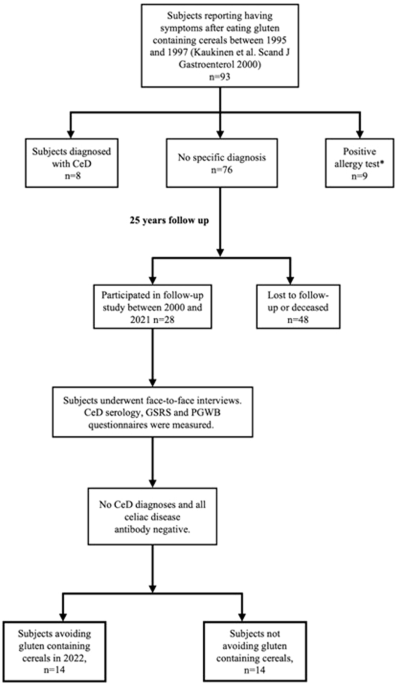
Patients and study design
The study was conducted at Tampere University and Tampere University Hospital in 2021. The present study was a follow-up to one carried out in the same center approximately 25 years earlier [10]. The previous study included 93 consecutive adults who experienced gastrointestinal symptoms after consuming gluten-containing cereal products such as wheat, rye, and barley [10]. In the study conducted in 1995–97 [10], participants underwent a variety of diagnostic investigations at baseline, including upper gastrointestinal endoscopy with duodenal biopsies, determination of CeD-associated genetics and serology, and comprehensive allergy testing (Fig. 1). Altogether 76 participants did not have CeD or wheat allergy and, according to standard treatment protocols at that time, they were advised to revert to normal gluten-containing diet. Those individuals whose contact details were available in the digital and population data services agency of Finland were invited to a follow-up study. Participants were invited to take part through up to two letters, in accordance with ethical approval. Those who responded by contacting the study nurse were subsequently scheduled for a visit at the Tampere Celiac Disease Research Center.

* Specific and total IgE antibodies for wheat were measured with additional prick and patch testing. After testing, the dermatologist deemed nine patients as having possible cereal allergies and started an open-label elimination diet for them [9]. CeD celiac disease.
During the study visit, participants were interviewed personally by a study nurse, and blood samples were taken for CeD antibody and laboratory measurements. The participants also completed specific questionnaires eliciting gastrointestinal symptoms and quality of life. The control groups comprised 128 untreated CeD patients collected from a prospectively maintained research database and 160 apparently healthy non-CeD individuals who were not on a gluten-free diet (GFD) and had no first-degree relatives with CeD [11]. The control patients were recruited through newspaper advertisements and via local and national CeD associations from different parts of Finland. “Untreated CeD” refers to individuals who had received a CeD diagnosis but were studied prior to diagnostic investigations and before starting a GFD. Following small-bowel mucosal biopsy findings of villous atrophy and crypt hyperplasia, these patients were placed on a GFD.
The study protocol and participant recruitment were approved by the Ethics Committee of the Pirkanmaa Hospital District. Written informed consent was obtained from all participants. The Declaration of Helsinki was followed throughout the study process.
Clinical data
The following demographic and clinical data were collected from all participants: Age, self-reported gender, body mass index, possible CeD diagnosis or other chronic disease after the first study in 1995–97, and presence and duration of possible persistent gastrointestinal symptoms. In addition, the possible avoidance of gluten-containing cereals was systematically assessed using a written questionnaire, with the responses reviewed by a study nurse. Participants were asked whether their reason for gluten avoidance was symptoms, assumed negative long-term health effects, or other factors, with the option to select multiple reasons. Adherence to a possible GFD was further categorized as strict, occasional lapses, or no diet. Participants were also asked to rate the ease of following the possible diet on a scale of 1–5, its impact on daily life (yes/no), the frequency of follow-up (never, occasionally, every 2–3 years, yearly), and the current follow-up method (doctor, nurse, remote contacts, or other). The gastrointestinal symptoms were systematically assessed to evaluate potential fulfillment of the Rome IV criteria for IBS [12].
Laboratory measurements
In this follow-up study, serum IgA-class tissue transglutaminase antibodies (TGA) were analyzed at the Tampere Celiac Disease Research Center by commercial assay (Celikey®, Phadia GmbH, Freiburg, Germany). Serum IgA-class endomysial antibody (EmA) titers were determined by indirect immunofluorescence using human umbilical cord as substrate [13]. In case of suspected IgA deficiency, the antibodies were measured in the IgG class. A serum dilution of 1 ≥ 5 for EmA was considered positive, and positive samples were further diluted up to 1:4000. Dynal SSP low-resolution DQ typing kit (Dynal AS, Oslo, Norway) was used to determine the presence of CeD-associated HLA DQ2 and DQ8 haplotypes [14]. Blood hemoglobin was measured with a commercial HemoCue Hb 201+ analyzer (Triolab Inc., Turku, Finland). Anemic values were defined based on sex-specific reference ranges: 117–155 in women and 134–167 in men [15].
Questionnaires
Gastrointestinal symptoms and health-related quality of life and well-being were assessed quantitatively using three validated and widely used questionnaires, including the Gastrointestinal Symptom Rating Scale (GSRS), the Psychological General Well-Being Index (PGWB), and the Rand 36-Item Short Form Health Survey (SF-36). GSRS and PGWB were available from both participants and controls, while scores from a separate Finnish population sample (n = 2060, 1133 women and 927 men) were used for comparison of gender-specific SF-36 values. Altogether 1529 of these were aged 18–64 years, and 513 were over 65 years old [16].
GSRS estimates self-experienced gastrointestinal symptoms with 15 separate questions utilizing a Likert scale from 1 to 7 points, higher scores denoting more severe symptoms [17, 18]. Independent questions are further combined into subcategories of diarrhea, indigestion, constipation, pain, and reflux. The results are given as mean scores for total and subdimension scores.
The PGWB questionnaire assesses psychological health and well-being with 22 separate items, which are further combined into subcategories of anxiety, depression, well-being, self-control, general health, and vitality [17, 19]. Each question is answered on a Likert-like scale from 1 to 6, and the results are summed for total and subdimension scores. Higher scores indicate better self-perceived health and well-being [17, 19].
The SF-36 evaluates various aspects of health and quality of life [20]. It comprises eight subcategories, including physical functioning, bodily pain, role limitations due to physical health problems, role limitations due to personal or emotional problems, emotional well-being, social functioning, energy/fatigue, and general health perceptions. The scores of each item may range from 0 to 100 points, with higher values indicating better health status.
Statistical analysis
Statistical analyses were conducted using IBM SPSS Statistics software (IBM Corp., Armonk, New York, USA). The results are given either as numbers of cases and percentages or as means with 95% confidence intervals (CI) or ranges. Visual inspection and Shapiro–Wilk and Kolmogorov–Smirnov tests were used to confirm the normality of the data. Independent samples T-test, Mann–Whitney U test, and chi-square were used for testing statistical significance when appropriate. In addition, a one-sample T-test was used when comparing the SF-36 scores of the study participants to the scores of the Finnish population sample. A two-tailed p-value < 0.05 was considered significant in all comparisons. A separate comparison analysis was conducted between those ingesting gluten-containing products and those avoiding them.
link