Study design and research objectives
In this study, we utilized a retrospective cohort study design to examine the outcomes of carpal tunnel release surgery, focusing on the perspectives and patient-reported experiences.
Our primary objective was to investigate these outcomes using the Boston Carpal Tunnel Questionnaire (BCTQ). Specifically, we aimed to measure the BCTQ scores before and after surgery, as well as determine the difference between the pre and postoperative scores.
Additionally, we sought to explore secondary outcomes, including the patients’ health profiles, medical comorbidities, disease presentations, and treatment modalities used. We then aimed to analyze these factors alongside the BCTQ score differences to identify any potential associations.
Participants and setting of the study
In this study, we examined a total of 681 patients who underwent carpal tunnel release surgery at two leading hospitals in Jordan over a period of five years, from January 2018 to December 2022. These patients were diagnosed with carpal tunnel syndrome through clinical examination, and confirmation of the diagnosis was achieved using electrodiagnostic studies.
Inclusion and exclusion criteria
The inclusion criteria for this study consisted of patients who underwent isolated open carpal tunnel release surgery between January 2018 and December 2022. However, patients who underwent combined procedures during the carpal tunnel release surgery, had endoscopic surgery, or underwent revision surgery were excluded from the study. Additionally, patients who refused to participate, had incomplete health records, had previous surgery on the operated hand, had previous hand trauma or fractures on the operated hand, or had systemic or peripheral neuropathies were also excluded from the study.
Study sample and sample size
A convenience sampling method was employed for patient recruitment in this study. The selected sample size of 681 patients was deemed sufficient for the purpose and objectives of our research and was determined based on several considerations. Firstly, a review of prior studies on this topic indicated that the majority of them utilized sample sizes that were comparable to, or smaller than, ours, thereby suggesting the adequacy of our chosen sample size. Moreover, the selection of 681 patients was also influenced by practical considerations, particularly the operational capacity and resources available within the two hospitals included in our study. By aligning our sample size with the feasibility and logistical capabilities of the study sites, we ensured the smooth and efficient conduct of data collection within the designated timeframe.
Outcome variable definitions
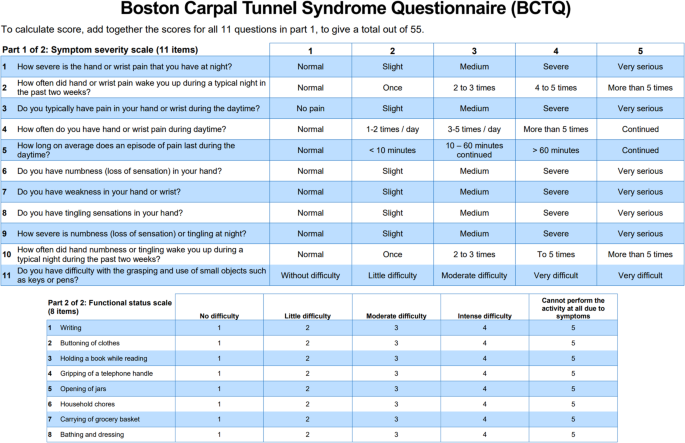
In this study, the primary outcome variable of interest was the Boston Carpal Tunnel Questionnaire (BCTQ). Levine et al. introduced the BCTQ in 199317, which includes two distinct sections.
The first section, known as the Symptom Severity Scale, is composed of 11 questions aimed at evaluating the severity of symptoms associated with carpal tunnel syndrome. Each question is scored on a scale of 1 to 5, where a lower score indicates a more favorable condition (1 indicates the least severe symptoms and 5 indicates the most severe).
The second section, the Functional Status Scale, assesses the patient’s functional status through 8 items, each scored similarly on a scale of 1 to 5. Here, a lower score again denotes a better condition (with 1 being the least impairment and 5 being the most impairment).
After completing the BCTQ, the average scores for both symptom severity and functional status scales were calculated. Our study included evaluating pre- and post-surgery BCTQ scores and determining the score differences, thus offering a comprehensive assessment of each patient’s condition.
Study protocol
We included patients diagnosed with carpal tunnel syndrome through clinical evaluation and confirmed with electrodiagnostic studies who underwent carpal tunnel release surgery between January 2018 and December 2022. Following the application of specific inclusion and exclusion criteria, 681 patients were deemed eligible for analysis. The surgical procedures were conducted by skilled orthopedic surgeons using a standardized technique at day-case surgery centers within two hospitals. All patients underwent assessment using the Boston Carpal Tunnel Questionnaire (BCTQ) within one week before surgery, and BCTQ was subsequently re-assessed postoperatively during clinic follow-up visits, interviews, and phone calls with patients.
Ethical consideration
This study received approval from the Institutional Review Board (IRB) at Jordan University’s Medical Research Office, with IRB number 2023/25,128. Prior to enrollment, all participants provided informed consent, and strict measures were implemented to safeguard patient confidentiality and privacy throughout the study. The research adhered to the Code of Ethics outlined by the World Medical Association (Declaration of Helsinki).
Data collection and analysis
The data collected in this study is organized into three main categories. Firstly, it includes an examination of patients’ health profiles and medical comorbidities, encompassing variables such as age, gender, smoking habits, and the presence of comorbidities. Secondly, the dataset contains information related to patients’ symptomatology, including aspects such as pain, paresthesia, and nocturnal symptoms. Lastly, the study incorporates patient-reported outcome variables, specifically the Boston Carpal Tunnel Questionnaire (BCTQ).
Data were collected and analyzed using the Statistical Package for Social Science (SPSS), version 23. The data collection process involved postoperative follow-up visits, phone calls, and authorized access to patients’ health records. Descriptive statistics were utilized to summarize patient characteristics and patient-reported outcomes (PROs), with a statistical significance level set at p < 0.05 to determine significant findings.
Quantitative variables such as age, weight, height, and BMI were analyzed using descriptive statistics, with means and standard deviations calculated. The relationship between mean differences in pre- and postoperative BCTQ scores was assessed using Student’s t-test.
Figure 1 illustrates the Boston Carpal Tunnel Questionnaire, and Fig. 2 presents a concise flowchart summarizing our study methodology.

(a) The first part of the BCTQ, the symptoms severity scale26,27. (b) The second part of the BCTQ, the functional status scale26,27.

Flowchart summary of the research methodology.
link